Mechanochemical synthesis of multi-site electrocatalysts as bifunctional zinc–air battery electrodes†
Abstract
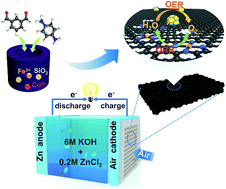
The sluggish kinetics of two crucial reactions, the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER), limit the practical application of rechargeable zinc–air batteries (ZABs). How to construct high-activity sites for the reversible ORR and OER together in the air cathode still remains challenging. Herein, by introducing Fe into Co-doped carbon, an efficient bifunctional ORR and OER electrocatalyst with multiple active sites of uniformly dispersed alloy nanoparticles, atomic-scale metal-N coordinated species and pyridinic-N groups has been successfully prepared by mechanochemical condensation and subsequent pyrolysis. Fe-doping into a Co-based system not only results in the formation of alloyed FeCo nanoparticles that greatly enhance the OER kinetics, but also helps increase the ratio of pyridinic-N and metal-N groups and promote electron transfer between the metal and N, significantly improving the ORR activity. DFT calculation reveals that the FeCo alloy is more energetically favorable than pure Co in the OER process through increasing the adsorption energy for OH and accelerating the dehydrogenation step. The ZABs assembled using the optimized electrocatalysts as the air cathode exhibit outstanding charge and discharge performance, large power density, and long cycling life, much better than those using precious metal catalysts Pt/C and RuO2. The present study presents a scalable mechanochemical strategy to synthesize low-cost carbon-based materials with well-defined multiple active sites, showing great promise in energy storage, adsorption, and catalysis.


 Please wait while we load your content...
Please wait while we load your content...