Metallovesicles as smart nanoreactors for green catalytic synthesis of benzimidazole derivatives in water†
Abstract
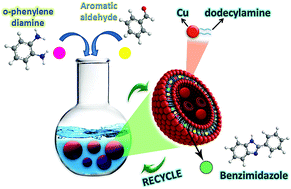
Metallovesicles are an emerging class of soft nanomaterials where spherical bilayer membranes, resulting from self-aggregation of amphiphilic metal complexes, amalgamate the advantages of metal specific catalytic properties and small hydrophobic cavities serving as nanoreactors. The confinement of substrates in these vesicle bilayers, on account of hydrophobic interactions in aqueous media, encourage their application in catalysis, particularly, where preclusion of organic solvents is of prime concern without compromising desired reaction rates and product yields. In the present work, novel amphiphilic metallosurfactant complex [Cu(C12H25NH2)2]Cl2 has been self-assembled to achieve spherical bilayer structures known as copper metallovesicles (CuMVs). DLS, TEM and FESEM analyses revealed the formation of spherical multivesicular vesicles in the size range 160–200 nm. The multivesicular structure of CuMVs was further supported by small angle X-ray scattering (SAXS) results. The as-synthesized CuMVs were further assessed for their potential as aqueous catalytic system for the synthesis of important therapeutic agents, benzimidazoles. The co-existence of hydrophobic reactants inside the metal-adorned vesicle bilayers, affords high product yield in short times. Facile synthesis of metallosurfactants, self-assembly to metallovesicles, aqueous reaction media, low metal concentration, low E-factor values, stability and recyclability of metallovesicles are the features that establish metallovesicular catalysis as a promising multifaceted approach for greener catalysis of benzimidazole synthesis and many other significant synthetic reactions.
- This article is part of the themed collection: Research presented at the International Conference on Emerging Advanced Nanomaterials (ICEAN) 2018


 Please wait while we load your content...
Please wait while we load your content...