Hydrides of early transition metals as catalysts and grain growth inhibitors for enhanced reversible hydrogen storage in nanostructured magnesium†
Abstract
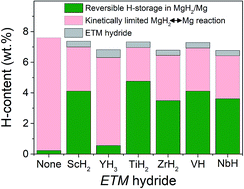
Magnesium is a remarkable hydrogen storage material due to its ability to reversibly absorb a high dihydrogen amount at affordable cost. However, its practical use is hampered by the high thermodynamic stability of the hydride and slow reaction kinetics. In this work, one-pot synthesis of nanostructured magnesium hydride with addition of 5 mol% of Early Transition Metals (ETM = Sc, Y, Ti, Zr, V, and Nb) as hydrogenation catalysts is accomplished by mechanochemistry under hydrogen gas. Structural and hydrogenation properties have been systematically analyzed to gain a deep understanding of the influence of ETMs on the hydrogenation properties of magnesium. The as-synthesized materials are nanocomposites of MgH2 and ETM hydrides (ScH2, YH3, TiH2, ZrH2, VH and NbH) with a crystallite size of ∼10 nm. All nanocomposites, but MgH2–YH3, have high reversible hydrogen storage (≥5 wt%) at 573 K thanks to catalytic effects induced by ETM hydrides leading to fast sorption kinetics. We here demonstrate that, on desorption, ETM hydrides can catalyze the recombination of hydrogen atoms. On absorption, formation of coherent interfaces between ETM hydrides and MgH2 favors nucleation of the latter. Moreover, for the peculiar case of TiH2, lattice mismatch between Mg and TiH2 hydride limits Mg grain growth, which preserves the fast absorption kinetics of the MgH2–TiH2 nanocomposite on cycling. Thus, the best H-cycling properties are found for the MgH2–TiH2 nanocomposite with a reversible capacity of 4.8 wt% after 20 H-cycles and the reaction time arbitrarily limited to 15 min.


 Please wait while we load your content...
Please wait while we load your content...