Visualizing percolation and ion transport in hybrid solid electrolytes for Li–metal batteries†
Abstract
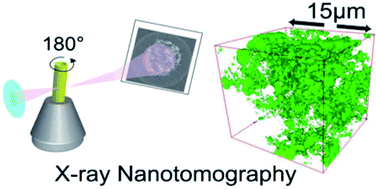
Hybrid solid electrolytes are composed of organic (polymer) and inorganic (ceramic) ion conducting materials, and are promising options for large-scale production of solid state lithium–metal batteries. Hybrid solid electrolytes containing 15 vol% Al-LLZO demonstrate optimal ionic conductivity properties. Ionic conductivity is shown to decrease at high inorganic loadings. This optimum is most obvious above the melting temperature of polyethylene oxide where the polymer is amorphous. Structural analysis using synchrotron nanotomography reveals that the inorganic particles are highly aggregated. The aggregation size grows with inorganic content and the largest percolating clusters measured for 5 vol%, 15 vol% and 50 vol% were ∼12 μm3, 206 μm3, and 324 μm3, respectively. Enhanced transport in hybrid electrolytes is shown to be due to polymer|particle (Al-LLZO) interactions and ionic conductivity is directly related to the accessible surface area of the inorganic particles within the electrolyte. Ordered and well-dispersed structures are ideal for next generation hybrid solid electrolytes.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...