Rate-independent and ultra-stable low-temperature sodium storage in pseudocapacitive TiO2 nanowires†
Abstract
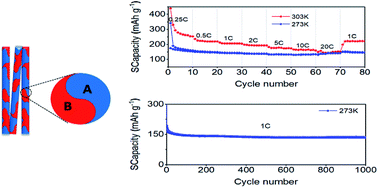
Sodium-ion batteries (SIBs) are faced with several challenges, including low capacity, short cycle life, and poor low-temperature performance. In this work, TiO2-B/anatase dual-phase nanowires are synthesized and applied as SIB anodes to address the above challenges. For the first time, we find the excellent Na-storage performance of the nanowire anode like rate-independent capacities and ultra-stable cycling stability at low temperature. Operando Raman spectroscopy shows that the nanowires are completely amorphized after cycling at 303 K; however, the TiO2-B phase of the dual-phase nanowires remains crystalline after cycling at 273 K. The different sodiation mechanisms at different temperatures result in a lower capacity but a more stable structure during cycling at 273 K than at 303 K. Kinetic analysis shows that the nanowire anode possesses an ultralow charge-transfer energy barrier and resistance with a higher apparent Na diffusion coefficient at 273 K than at 303 K during desodiation, which significantly enhances the Na+ intercalation pseudocapacitive process at low temperature. The synergy between the structural transition and diffusion kinetics leads to rate-independent and ultra-stable Na-storage performance at low temperature. This work provides new perspectives for the understanding and design of low-temperature SIBs with high rate capability and long cycle life.


 Please wait while we load your content...
Please wait while we load your content...