Nanostructured Co-based bifunctional electrocatalysts for energy conversion and storage: current status and perspectives
Abstract
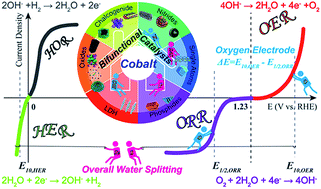
Electrocatalytic hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and oxygen reduction reaction (ORR) are three typical reactions in energy conversion devices, such as water electrolyzers, metal–air batteries and fuel cells. However, the sluggish kinetics of the HER/OER/ORR and their dependency on noble-metal-based catalysts (e.g. Pt, Ir and Ru) hinder the large-scale commercial applications of these devices. Hence, the development of low-cost, efficient, stable and scalable electrocatalysts, especially bifunctional HER/OER or OER/ORR electrocatalysts that could simultaneously catalyze HER/OER or OER/ORR, is highly desired but full of great challenges. To date, non-noble cobalt (Co)-based materials, including cobalt oxide, cobalt phosphides, cobalt chalcogenides (sulfide and selenides), Co-included layered double hydroxides, Co–N–C, Co-based single atoms and their composites, have been widely investigated as bifunctional electrocatalysts owing to their innate electrochemical capabilities and structural adjustability by mixing, doping or combining with other materials, such as carbon materials. In this review, the fundamental mechanisms of the bifunctional electrocatalysis of ORR/OER and OER/HER are introduced and analyzed, and recent progress in the design, synthesis and emerging applications of Co-based materials as bifunctional electrocatalysts for ORR/OER or OER/HER in alkaline electrolytes are summarized and discussed. Finally, the strategies and perspectives for designing novel highly efficient bifunctional cobalt-based electrocatalysts are analyzed. This analysis is expected to highlight the challenges faced by bifunctional noble-metal-free electrocatalysts for the study and development of next-generation electrocatalysts for the HER/ORR/OER.
- This article is part of the themed collection: Recent Review Articles


 Please wait while we load your content...
Please wait while we load your content...