Continuous oxygen vacancy engineering of the Co3O4 layer for an enhanced alkaline electrocatalytic hydrogen evolution reaction†
Abstract
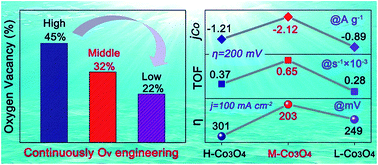
Precisely tailoring the oxygen vacancies (Ov) of Co oxide based electrocatalysts could be highly essential for boosting hydrogen evolution reaction (HER) performance in alkaline media. Here, a rapid synthesis of an Ov-engineered Co3O4 layer on Ni foam has been demonstrated through a solid-phase melting strategy, where the Ov content in the Co3O4 layer can be adjusted from 52% to 74% simply by changing melting temperature. After HER testing, this content is further adjusted to 22–45% with the same trend. Remarkably, the Co3O4 layer with a medium Ov content (32%) gives the highest mass activity of −2.12 A g−1 at an overpotential of 200 mV for the alkaline electrocatalytic HER. Moreover, it only requires an ultrasmall overpotential of 203 mV to achieve −100 mA cm−2, outperforming all reported Co3O4 electrocatalysts so far. The medium Ov content in the Co3O4 electrocatalyst could enhance reaction kinetics by optimizing catalytic intermediate adsorption during the alkaline HER. This feasible tactic to precisely control Ov may provide an avenue for exploring other advanced electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...