Theoretical calculations, molecular dynamics simulations and experimental investigation of the adsorption of cadmium(ii) on amidoxime-chelating cellulose
Abstract
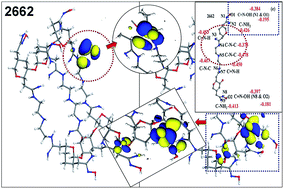
Amidoxime-chelating cellulose (ACCS) was obtained through comprehensive modification, i.e. alkalization, etherification, amination and chelating; then, the adsorption of cadmium(II) was investigated by batch experiments, characterization, computational theoretical calculations and molecular dynamics simulations. Isothermal adsorption showed that the Freundlich equation described the adsorption data well at pH 5.0 and 7.0, whereas the Langmuir model was in good accordance at pH 1.0 and 3.0. As confirmed, ACCS exhibited the best Cd(II) adsorption capacity (134.13 mg g−1) at pH 5.0 and even retained considerable adsorption performance under extremely acidic conditions. Additionally, it maintained excellent adsorbability and little weight loss after 5 cycles. Further, the adsorption structure of 2662 type ACCS was determined by quantum molecular (QM) and molecular orbital (MO) investigations. On the basis of this structure, the amidoxime group (–C(NH2)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NOH) was proved to be key in the adsorption process by Mulliken charge analysis and molecular dynamics simulations. Furthermore, the binding energy values clearly indicated that Cd(II) favors binding with C–NH2 over C
NOH) was proved to be key in the adsorption process by Mulliken charge analysis and molecular dynamics simulations. Furthermore, the binding energy values clearly indicated that Cd(II) favors binding with C–NH2 over C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–OH in the amidoxime group. In summary, our interpretation of the overall adsorption mechanism was well quantified by efficient calculations and simulations.
N–OH in the amidoxime group. In summary, our interpretation of the overall adsorption mechanism was well quantified by efficient calculations and simulations.


 Please wait while we load your content...
Please wait while we load your content...