Organic–inorganic hybrids of Fe–Co polyphenolic network wrapped Fe3O4 nanocatalysts for significantly enhanced oxygen evolution†
Abstract
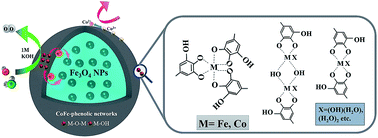
The design of organic–inorganic hybrid nanostructures with carefully balanced properties is a promising approach for constructing advanced electrocatalysts using earth abundant elements. Here we describe hybrid electrocatalysts that have strongly coupled metal (Co–Fe) polyphenolic networks (MPNs) and Fe3O4 nanoparticles for catalyzing the oxygen evolution reaction (OER) with exceptional activity. These catalysts were produced by a tannic acid (TA) mediated conversion process that selectively converts the surface layers of pre-synthesized zeolitic imidazole framework-67 (ZIF-67)@Fe3O4 core–shell nanoparticles to uniformly wrapped organic MPN layers. The MPN layers are composed of metal–phenolic complexes that are rich in molecular metal hydro(oxy)oxide (M–OH, M–O–M) centers that not only increase the electrochemical surface area by approximately four times but also serve as highly active sites for the OER. As a result of the structural and compositional benefits, the hybrid catalysts only require an overpotential of 260 mV to produce oxygen at 10 mA cm−2 and have a Tafel slope of only 33.6 mV dec−1 in alkaline electrolytes, which are the lowest when compared with pristine ZIF-67@Fe3O4, Fe3O4 and the benchmark IrO2 catalysts.


 Please wait while we load your content...
Please wait while we load your content...