Insight into the role of Ni–Fe dual sites in the oxygen evolution reaction based on atomically metal-doped polymeric carbon nitride†
Abstract
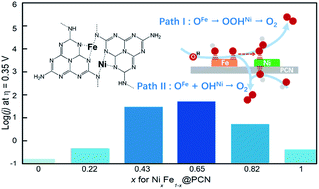
The oxygen evolution reaction (OER) plays a critical role in efficient conversion and storage of renewable energy sources, whereas the active sites for the most representative electrocatalysts, Ni–Fe compounds, remain under debate. In this work, we have developed polymeric carbon nitride (PCN) with atomically dispersed N-coordinated Ni–Fe sites to investigate the OER process. The Ni–Fe dual sites consist of adjacent Ni and Fe atoms coordinated with N atoms in the PCN matrix. NiFe-codoped PCN exhibits higher electrocatalytic activity than monometal-doped catalysts, showing a lower overpotential (310 mV at 10 mA cm−2) and smaller Tafel slope (38 mV dec−1) in 1 M KOH, indicating that Ni–Fe dual-metal sites significantly favor the OER process. According to density functional theory calculations based on the oxidized-NiFe@PCN model, it was found that adjacent Ni and Fe atoms co-participate in the OER process for NiFe-codoped PCN, leading to a much lower energy barrier (0.10 or 0.22 eV for U = 1.58 V), while the effect of electronic modification of the single metal active sites by the other component (Ni sites by Fe sites or vice versa) contributes less to activity enhancement, thus leading to a rational explanation on the synergistic effect of the NiFe-based OER catalysts.


 Please wait while we load your content...
Please wait while we load your content...