Microwave-assisted ultrafast synthesis of adjustable bimetal phosphide/graphene heterostructures from MOFs for efficient electrochemical water splitting†
Abstract
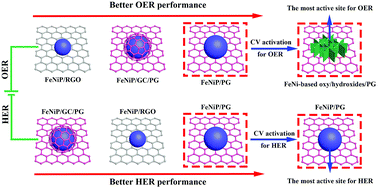
Herein, we develop a novel ultrafast microwave-assisted thermal conversion route to tune the heterostructure of transition metal phosphide/graphene composites for oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) activity enhancement. With this method, transition metal phosphide/graphene composites with adjustable heterostructures including transition metal phosphide/P-doped graphene and core@shell transition metal phosphide@graphitized carbon/P-doped graphene could be obtained by using metal organic framework (MOF)/graphene as a precursor within 20 s. In contrast, traditional programmed heat treatment only results in transition metal phosphide/non-doped graphene. We reveal for the first time that transition metal phosphides and their derived oxy/hydroxides, rather than the long thought graphitized carbon covering the surface of transition metal phosphides, are the most active sites for the HER and OER, respectively. Meanwhile, P-doping in graphene could highly improve its electrocatalytic performance while graphitized carbon encapsulation might impair its performance by separating it from the electrolyte. Therefore, bimetal phosphide (FeNiP)/P-doped graphene was determined as the most active metal phosphide/graphene heterostructure and it could deliver a very low overpotential of about 229 and 173 mV at 10 mA cm−2 in alkaline media for the OER and HER, respectively. Furthermore, a high-performance overall water splitting cell that could achieve a current density of 10 and 50 mA cm−2 at 1.58 and 1.74 V, respectively, was successfully constructed by using FeNiP/P-doped graphene as both cathode and anode.


 Please wait while we load your content...
Please wait while we load your content...