Exploration of the sodium ion ordered transfer mechanism in a MoSe2@Graphene composite for superior rate and lifespan performance†
Abstract
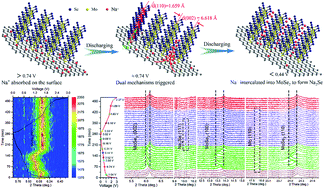
Two-dimensional structured materials have attracted much attention and are commonly employed as active materials for sodium ion batteries owing to their inherent advantages. The reaction mechanisms behind sodium ion storage and diffusion pathways in a composite of MoSe2@graphene were monitored and explored by the in situ synchrotron technique. Based on nanocages in the MoSe2 structure and efficient sodium ion pre-location on the extensive surfaces of graphene, the combination of a sodium ion storage mechanism and an ordered transfer mechanism is proposed in different voltage windows. First, numerous sodium ions are pre-located on the surface by an adsorption reaction. In the second step, all the pre-located sodium ions are immediately inserted into MoSe2 nanosheets, which corresponds to the intercalation reaction mechanism. In the final step, the intercalated sodium ions will undergo a conversion reaction with MoSe2. Meanwhile, numerous nanocages resulting from sufficient deficiency of MoSe2 and layer dislocation will be beneficial to the capacity enhancement and rate capability. The prior adsorption reaction can efficiently improve the rate capability as well. Benefitting from these merits, the composite exhibits a high specific capacity of 640 mA h g−1 and robust rate performance. The cycling lifetime is also increased as well, with a capacity retention of ∼85% over 1000 cycles at a current density of 1.0 A g−1. Therefore, this composite shows promising potential for application in future sodium ion batteries.


 Please wait while we load your content...
Please wait while we load your content...