Pore environment engineering in metal–organic frameworks for efficient ethane/ethylene separation†
Abstract
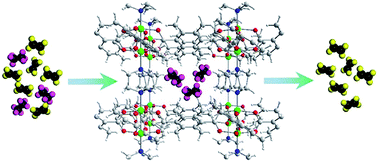
Selective adsorption of trace amounts of C2H6 from bulk C2H4 is a significantly important and extremely challenging task in industry, which requires an adsorbent with specific pore properties. Herein, we describe a strategy for adjusting the pore environment of metal–organic frameworks (MOFs) by introducing different amounts of methyl groups in the channel to enhance the guest–host interaction between C2H6 and the framework. To prove this concept, 2,3,5,6-tetramethylterephthalic acid (TMBDC) was deliberately added to a microporous MOF, Ni(BDC)(DABCO)0.5, affording a series of mixed-ligand materials, Ni(BDC)1−x(TMBDC)x(DABCO)0.5 (x = 0, 0.2, 0.45, 0.71, 1), having different pore environments. Significantly, these mixed-ligand materials demonstrated improved performance in terms of the adsorption capacity of C2H6 and C2H4 with an unprecedented C2H6 uptake of 2.21 mmol g−1 for Ni(TMBDC)(DABCO)0.5 at 0.0625 bar and 298 K. With the best theoretical C2H6/C2H4 selectivity predicted by IAST, Ni(TMBDC)(DABCO)0.5 exhibited effective separation of C2H6/C2H4 (1/15, v/v) and great recyclability in five consecutive adsorption/desorption cycles throughout the breakthrough experiment.


 Please wait while we load your content...
Please wait while we load your content...
