Breaking the volcano-plot limits for Pt-based electrocatalysts by selective tuning adsorption of multiple intermediates†
Abstract
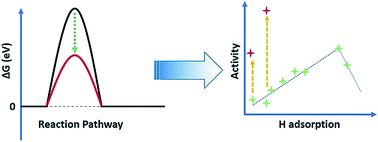
The development of heterogeneous electrocatalysts with enhanced activity as compared to conventional benchmarks is a highly important but challenging task. One of the main reasons is that the performance of current catalysts is limited by a ‘volcano plot’, which defines the relationship between the activities of a family of catalysts and a series of specific descriptors (e.g., adsorption ability of reaction intermediate(s)). Here, using the alkaline hydrogen evolution reaction as an example, we propose a strategy to break such activity limits. By building up volcano plots for a group of Pt alloys, we found that some “unique” samples did not follow the trend and possessed higher activities beyond the volcano top. Our thermodynamic and kinetic measurements revealed that dealloying can break the poorly known scaling relationship between the binding energies of *H and *OH, and thus, selectively optimize the adsorption properties toward *H and *OH reaction intermediates on Pt surfaces and boost the overall activity. This provides a new insight into the aforementioned activity limits, which can be avoided by regulating the adsorption behaviors of two or more intermediates.


 Please wait while we load your content...
Please wait while we load your content...