Investigation of the self-discharge behaviors of the LiMn2O4 cathode at elevated temperatures: in situ X-ray diffraction analysis and a co-doping mitigation strategy†
Abstract
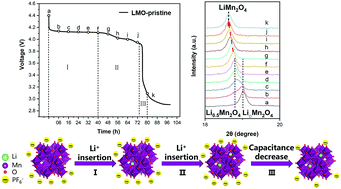
The practical application of lithium-ion batteries at elevated temperatures is significantly hampered by their poor cyclabilities and self-discharge behaviors. In this study, we investigated real-time structural evolution of the lithium manganese oxide cathode (LiMn2O4, LMO) in the idle charged state as well as the origin of the self-discharge process via in situ X-ray diffraction analysis. Moreover, we proposed a facile and scalable co-doping strategy for the incorporation of Al3+, F− and PO43− into the LMO spinel structure; in addition, the thermal stability of the LMO samples was ameliorated by suppressing the parasitic formation of a Li-rich phase of LMO upon long-term cycling. When evaluated at room temperature, the modified LMO exhibits the impressive areal mass loading of up to 17.8 mg cm−2, the highest areal capacities of ∼1.4 mA h cm−2 at 5C, robust cyclability even after 1000 charge/discharge cycles, lower polarization voltage and significant mitigation of the self-discharge process at the elevated temperature of 55 °C; thus, this low-cost scalable modification strategy makes the LMO cathode commercially feasible for high-temperature applications.


 Please wait while we load your content...
Please wait while we load your content...