Influence of fluorination on CO2 adsorption in materials derived from fluorinated covalent triazine framework precursors†
Abstract
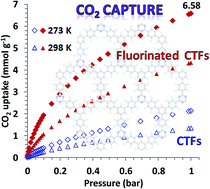
Ultra-nanoporous materials derived from fluorinated covalent triazine frameworks (CTFs) have been developed for highly efficient CO2 capture. A CO2 uptake capacity of 6.58 mmol g−1 at 273 K, 1 bar (2.45 mmol g−1 at 0.15 bar) is achieved. The excellent performance is due to the presence of ultra-micropores (0.6–0.7 nm) that tightly fit CO2 and strong electrostatic interactions from the residual fluorine atoms within the framework. Both molecular simulation and deep learning study predict that CTFs with a F content of ∼4.8 wt% and pore size distribution around ∼0.7 nm can give rise to the highest CO2 uptake capacity.


 Please wait while we load your content...
Please wait while we load your content...