Achieving highly efficient CO2 to CO electroreduction exceeding 300 mA cm−2 with single-atom nickel electrocatalysts†
Abstract
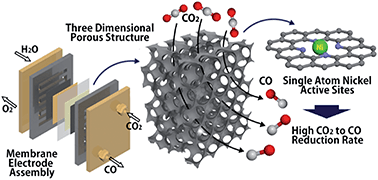
The electrochemical reduction of carbon dioxide (CO2) to value-added products is a promising approach to reduce excess CO2 in the atmosphere. However, the selective reduction of CO2 in aqueous electrolytes has been challenging owing to a competing hydrogen evolution reaction occurring in aqueous electrolytes. In this study, single atom nickel and nitrogen doped three-dimensional porous carbon catalysts are developed for the selective production of carbon monoxide (CO) from CO2. The catalysts exhibit high CO selectivity with over 99% faradaic efficiency at −0.8 V vs. the reversible hydrogen electrode (RHE), and achieve a high current density of over 50 mA cm−2 at −1.0 V vs. RHE in a bicarbonate electrolyte. To further improve the CO2 reduction rate, the accessibility of CO2 to the catalysts was enhanced by directly supplying gaseous CO2 to the surface of the catalysts. The catalysts were deposited between a gas diffusion layer and an ion exchange membrane to form a membrane electrode assembly (MEA). Benefiting from the high concentration of CO2 over the catalyst surfaces and the three-dimensional structure of the catalysts, a high CO production rate exceeding 300 mA cm−2 with 99% faradaic efficiency can be achieved.
- This article is part of the themed collection: International Year of the Periodic Table : Single Atoms as Active Catalysts


 Please wait while we load your content...
Please wait while we load your content...