Conductive metal–organic framework nanowire arrays for electrocatalytic oxygen evolution†
Abstract
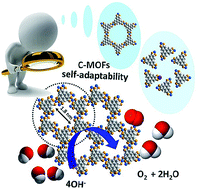
The design and construction of efficient electrode materials are significant for electrochemical energy conversion and storage technologies. The oxygen evolution reaction (OER) is a key process in water splitting devices and metal–air batteries. Herein, we report conductive metal–organic framework (C-MOF) nanowire arrays on carbon cloth as a promising electrocatalyst for OER. The pyrolysis-free C-MOF electrocatalyst can maintain intrinsic molecular active sites in the MOFs. The as-prepared electrode possesses overpotentials of ∼213 and 300 mV at 10 and 150 mA cm−2 and long-term stability in 1 M KOH, respectively. Control experiments and Fourier-transform extended X-ray absorption fine structure (EXAFS) and Mössbauer spectra indicate that the Fe doped in the Ni-based MOFs may serve as highly effective OER active sites. Density functional theory (DFT) calculations reveal an unusual self-adaptable property of the Fe active sites, which enables the OER intermediates to generate additional hydrogen bonds with the neighboring layer, thus lowering the free energy in the OER process. Our findings may provide an alternative method for developing MOF electrocatalysts in frontier potential applications.


 Please wait while we load your content...
Please wait while we load your content...