Acid induced conversion towards a robust and lithiophilic interface for Li–Li7La3Zr2O12 solid-state batteries†
Abstract
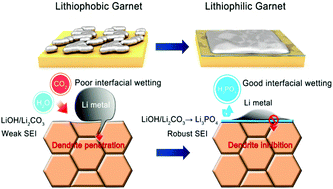
Solid-state lithium batteries (SSBs) promise high energy and power densities, as well as enhanced safety, owing to the use of Li metal and nonflammable solid-state electrolytes. In particular, cubic garnet-type Ta-substituted Li7La3Zr2O12 (LLZTO) is known for its high ionic conductivity and stability against Li metal. However, poor interfacial contact of the Li/LLZO interface is an obstacle that has come to light in the face of more demanding applications. Herein, to tackle this problem, a novel and facile strategy is demonstrated using H3PO4 to react with the Li2CO3/LiOH passivation layer of the LLZTO surface to form a uniform Li3PO4 modification layer. The Li3PO4 layer not only promotes interfacial wettability by reducing the significant difference of the surface energy, but also produces a robust solid electrolyte interphase (SEI) to inhibit lithium dendrite penetration. The symmetric cell exhibits a dramatically reduced interfacial resistance of 7.0 Ohm cm2 and ultrastable galvanostatic cycling, over 1600 hours at 0.1 mA cm−2 and over 450 hours at 0.5 mA cm−2. Besides this, hybrid solid-state full cells matched with different cathodes also displayed excellent electrochemical performances. LiFePO4 exhibited stable cycling (∼150 mA h g−1) and a good rate performance. In particular, for a S cathode, the coulombic efficiencies remained higher than 99% over 200 cycles. The use of a ternary NCM523 cathode also confirmed the anodic interfacial stability up to high voltage (4.5 V). Therefore, this facile strategy provides a stepping stone for future applications of solid-state Li-ion and Li–S batteries.


 Please wait while we load your content...
Please wait while we load your content...