Liquid crystalline lithium-ion electrolytes derived from biodegradable cyclodextrin†
Abstract
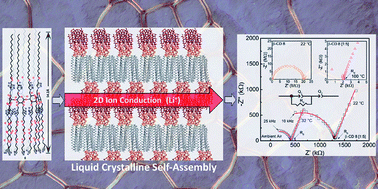
The first family of lithium-ion electrolytes based on thermotropic liquid crystalline (LC) cyclodextrin (CD) is reported. The new electrolytes consist of composites of self-assembling and environment-friendly amphiphilic β-CDs with lithium bistrifluoromethanesulfonimidate (LiTFSI). The unique geometry of the CD scaffold allows for efficient control of the amphiphilicity of the molecule through chemical derivatizations. The introduction of non-polar alkyl chains at the primary face of CD and multiple short and polar nitrile-terminated tetraethylene glycol chains at the other face provides a new class of LC materials capable of self-organization into homeotropically-aligned smectic mesophases, which retain long-range order over a wide temperature window. The assembly of numerous ethoxy groups at the secondary face of CD serves as an elegant pre-organization of the ion-chelating groups to form two-dimensional ion-conducting pathways, facilitating the directional motion of metal ions. Studies of several LC composites with LiTFSI have revealed promising thermal stability and ionic conductivity. Solid-state NMR studies have uncovered an activation energy of 0.14 eV for lithium diffusion, while cyclic voltammetry confirmed the composites remain electrochemically stable over a potential range of up to ∼3 V versus Li/Li+. These results demonstrate the great potential of this new class of organic electrolytes for different metal ions.


 Please wait while we load your content...
Please wait while we load your content...