The doping of phosphorus atoms into graphitic carbon nitride for highly enhanced photocatalytic hydrogen evolution†
Abstract
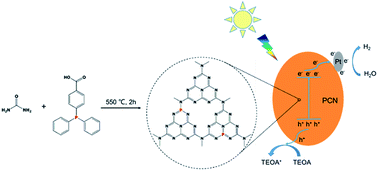
Graphitic carbon nitride (g-C3N4) is a promising candidate for visible-light-driven photocatalytic water splitting. However, some inherent drawbacks of g-C3N4, such as poor charge separation and transfer efficiencies, limited optical absorption and a low surface area, restrain its performance in photocatalytic hydrogen evolution. To solve these problems, in this study, a series of phosphorus (P) doped g-C3N4 (PCN) samples are prepared by direct thermolysis of a mixture of urea and 4-(diphenylphosphino)benzoic acid (4-DPPBA). P atoms have been successfully introduced into the framework of g-C3N4 by forming P–N bonds. The as-obtained samples are investigated for photocatalytic water splitting under visible light. Results reveal that the optimal PCN sample exhibits a high hydrogen evolution rate of 2610.80 μmol h−1 g−1 (λ ≥ 420 nm) in the presence of a sacrificial agent, which is almost 10 times higher than that of bare g-C3N4 (265.00 μmol h−1 g−1). This is mainly ascribed to the fact that electron-rich P atoms, acting as an electron donor, efficiently trap photogenerated holes and substantially suppress the recombination of charge carriers. Besides, the introduction of P atoms modifies the surface properties, narrows the band gap and increases the electrical conductivity. A possible mechanism is proposed for the photocatalytic reaction over PCN samples. Moreover, the PCN samples maintain high photocatalytic activity and chemical stability after recycling experiments, making them promising materials with broad application prospects in solar-to-fuel conversion.


 Please wait while we load your content...
Please wait while we load your content...