Pt-embedded in monolayer g-C3N4 as a promising single-atom electrocatalyst for ammonia synthesis†
Abstract
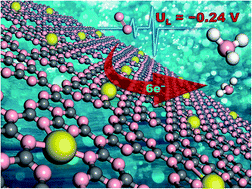
The development of advanced electrocatalysts for ambient NH3 production is increasingly attractive but severely plagued by low activity and poor selectivity. Herein, we reported first-principles results to explore the potential of transition metal (Sc– Cu, Mo, Ru, Rh, Pd, Re, Ir and Pt) atoms embedded in monolayer g-C3N4 as efficient single-atom electrocatalysts for NH3 production. We found that embedding single Pt atoms in g-C3N4 enables robust stability and good electrical conductivity and particularly results in excellent activity (with a low limiting potential of −0.24 V) at room temperature. Furthermore, the dominant N2 adsorption over that of H atoms suggests superior NH3 selectivity. Mechanistic analyses disclosed that single Pt atoms and g-C3N4 work in concert to address the significant deviation from the nitrogen linear scaling relationships, synergistically providing highly active sites for ambient electrochemical NH3 synthesis.


 Please wait while we load your content...
Please wait while we load your content...