Preparation and characterization of crosslinked poly(vinylimidazolium) anion exchange membranes for artificial photosynthesis†
Abstract
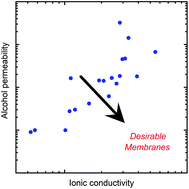
The interrelated nature of material properties in ion exchange membranes, such as ion exchange capacity and water uptake, frustrates the systematic study of how membrane chemistry and structure affect the transport of water, ions, and uncharged solutes in the membrane. Herein, we describe the preparation of a series of crosslinked poly(vinylimidazolium) anion exchange membranes by UV-photopolymerization of difunctional (i.e., crosslinking) and monofunctional (i.e., non-crosslinking) monomers, in which the ion exchange capacity and crosslink density may be independently controlled. Ion exchange membranes used in artificial photosynthesis (solar-driven CO2 reduction) devices must permit the transport of electrolyte ions and minimize the crossover of CO2 reduction products (e.g., alcohols) between electrodes. The water content, methanol (CO2 reduction product) permeability, and ionic conductivity of the membranes were evaluated. Ionic conductivity and methanol permeability were increased by reducing crosslink density or increasing solvent content in the prepolymerization solvent mixture. For all prepared membranes, methanol permeability was directly correlated with water volume fraction in the membrane. Minimizing the water volume fraction is critical to the design of membranes with low permeability to CO2 reduction products.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...