A comparative kinetic study of redox mediators for high-power lithium–oxygen batteries†
Abstract
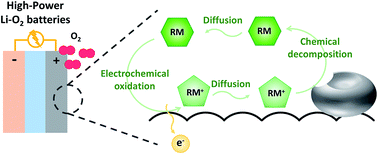
The use of redox mediators (RMs) effectively reduces the high polarization of lithium–oxygen batteries as RMs mediate the electrochemical formation and decomposition of discharge products. Since the electrochemical reactions are mediated by RMs, the power capability of the system would be critically dependent on the intrinsic kinetic properties of the RM in mediating the reaction. Herein, we performed a comparative kinetic study on several reported RMs of the oxygen evolution reaction by probing the RM-assisted charging process with respect to the rate of chemical decomposition of the discharge products and the diffusivity of the RMs in controlled lithium–oxygen cells. It was found that the overall kinetics of RMs had a positive correlation with the redox potential of the RM, and multi-redox RMs could display distinct properties depending on the oxidation states. Among the investigated RMs, DMPZ2+ (5,10-dihydro-5,10-dimethylphenazine) exhibited highest reaction rate for lithium peroxide decomposition, whereas TEMPO+ (2,2,6,6-tetramethyl-1-piperidinyloxy) showed highest mass diffusion rate. In addition, the choice of electrolytes is shown to significantly affect the rate capability of the RM-assisted charging and thus must be carefully considered. This study suggests the importance of understanding the kinetics of RMs and provides guidelines for achieving an optimized RM/electrolyte combination to realize high-power lithium–oxygen batteries.


 Please wait while we load your content...
Please wait while we load your content...