Room temperature demonstration of a sodium superionic conductor with grain conductivity in excess of 0.01 S cm−1 and its primary applications in symmetric battery cells†
Abstract
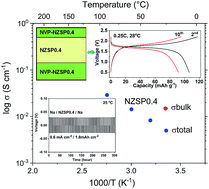
The lack of suitable candidate electrolyte materials for practical application limits the development of all-solid-state Na-ion batteries. Na3+xZr2Si2+xP1−xO12 was the very first series of NASICONs discovered some 40 years ago; however, separation of bulk conductivity from total conductivity at room temperature is still problematic. It has been suggested that the effective Na-ion conductivity is ∼10−4 S cm−1 at room temperature for Na3+xZr2Si2+xP1−xO12 ceramics; however using a solution-assisted solid-state reaction for preparation of Na3+xZr2Si2+xP1−xO12, a total conductivity of 5 × 10−3 S cm−1 was achieved for Na3.4Zr2Si2.4P0.6O12 at 25 °C, higher than the values previously reported for polycrystalline Na-ion conductors. A bulk conductivity of 1.5 × 10−2 S cm−1 was revealed by high frequency impedance spectroscopy (up to 3 GHz) and verified by low temperature impedance spectroscopy (down to −100 °C) for Na3.4Zr2Si2.4P0.6O12 at 25 °C, indicating further the potential of increasing the related total conductivity. A Na/Na3.4Zr2Si2.4P0.6O12/Na symmetric cell showed low interface resistance and high cycling stability at room temperature. A full-ceramic cell was fabricated and tested at 28 °C with good cycling performance.
- This article is part of the themed collection: Editor’s Choice: Solid-state ion conductors


 Please wait while we load your content...
Please wait while we load your content...