Tuning the electrocatalytic properties of a Cu electrode with organic additives containing amine group for CO2 reduction†
Abstract
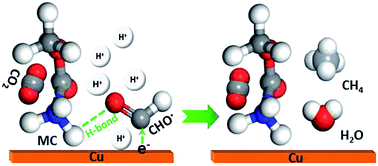
The conversion of CO2 into value-added chemicals by employing renewable energy via an electrochemical process has received increasing attention. However, the low reaction rate and efficiency, as well as poor selectivity, have limited its further development. The low solubility of CO2 in an aqueous solution is one of the main factors that limit the reaction rate and selectivity of the electroreduction of CO2 (ERC) at a high overpotential. In this study, an organic additive containing an –NH2 group (methyl carbamate, MC) was incorporated into the ERC electrolyte for the first time. By utilizing the affinity interaction between a Lewis acid (the –NH3+ group formed by protonation of the –NH2 group) and a Lewis base (CO2), the ERC reaction rate was increased by 17%, and the faradaic efficiency for the production of CH4 reached 81.6%, which represented a large increase of 34% and surpassed that of most reported Cu electrodes. Furthermore, the partial current density for the production of CH4 was 31 mA cm−2 at −2.13 V (vs. RHE), which represents an increase of 30% in comparison with that in the blank system. In addition, the competitive hydrogen evolution reaction was substantially inhibited, and the decline in faradaic efficiency increased from 38.5% to 48.1% when the potential decreased gradually from −1.63 V to −1.98 V (vs. RHE). DFT calculations further indicate that the –NH3+ group can significantly promote the adsorption of CO* and CHO* on the Cu surface and increase the surface coverage of both intermediates. CHO* can be further stabilized by the –NH3+ group via the facilitation of partial electron transfer from Cu to C and the formation of H-bonds, which is favorable for further hydrogenation to form hydrocarbons. These results provide a simple and powerful way to increase the reaction rate and product selectivity of the reduction of CO2.


 Please wait while we load your content...
Please wait while we load your content...