Physicochemical implications of alkoxide “mixing” in polyoxovanadium clusters for nonaqueous energy storage†
Abstract
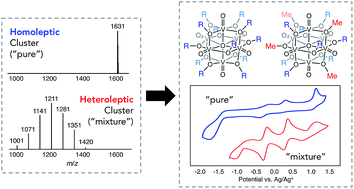
Identification of suitable charge carriers is essential for the success of nonaqueous energy storage systems. Thus, elucidation of structural parameters which affect the physicochemical properties of a molecule in organic solvent is of critical importance. Here, we continue our work related to the development of polyoxovanadium-alkoxide clusters for nonaqueous energy storage through the synthesis and subsequent analysis of a series of “mixed”-alkoxide clusters, [V6O7(OR)12−x(OCH3)x] (R = C2H5, C3H7, C4H9, C5H11, C6H13). We determine that the presence of a mixture of bridging-alkoxides enhances both the physical and electrochemical behaviour of the clusters in nonaqueous energy storage schematics, evidenced by increased solubility and electron-transfer kinetics. These results provide insight into the consequences of using a classically “impure” system, revealing how synergistic behaviour of a mixture of compounds can yield improved function over that of a pure solution.


 Please wait while we load your content...
Please wait while we load your content...