Hierarchical structural designs of ion exchange membranes for flow batteries†
Abstract
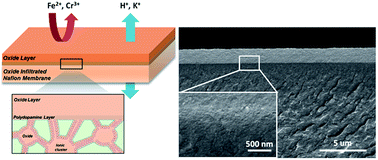
Membranes for flow batteries need to have both high ionic conductivity and selectivity. The selectivity is particularly important for systems involving two different species, such as Fe/Cr, since any crossover is irreversible. However, most membranes suffer from a trade-off between these two properties. We addressed this issue by fabricating a composite cation exchange membrane with a hierarchical structure to ensure mechanical stability, conductivity, and selectivity. The membrane featured a dense, crack-free tungsten oxide coating layer on Nafion that also penetrated into the Nafion's hydrophilic, ionic cluster regions. The oxide inside the polymer was synthesized in situ in the hydrophilic regions, while the sol–gel method was used to coat the oxide on the polymer. The oxide inside Nafion reduced water uptake and swelling ratio, which are essential to ensure the adhesion of the top oxide layer on Nafion. The top dense oxide layer served as a highly selective membrane for cation transport. Maintaining comparable conductivities, the membrane showed that the permeabilities for Fe(II) and Cr(III) ions decrease by an order of magnitude as compared to those for Nafion (from 2.66 × 10−7 cm2 s−1 and 8.84 × 10−8 cm2 s−1 to 2.31 × 10−8 cm2 s−1 and 8.34 × 10−9 cm2 s−1, respectively). Noticeably, the oxide coating layer showed three orders of magnitude lower permeability than Nafion. When applied in a lab-scale Fe/Cr flow battery, the cycling stability significantly improved due to the much reduced crossover. This new membrane design can be extended to other polymer membranes and holds promise for applications in a variety of flow batteries.


 Please wait while we load your content...
Please wait while we load your content...