Composite K2Mo4O13/α-MoO3 nanorods: sonochemical preparation and applications for advanced Li+/Na+ pseudocapacitance†
Abstract
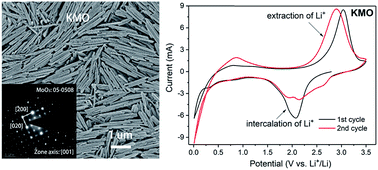
The proposal of pseudocapacitive materials breaks the barriers between batteries and capacitors, allowing for the possible achievement of a balanced energy-power performance. However, the limited reserve of lithium restricts the practical applications of pseudocapacitive materials in lithium-based systems, whereas sodium resources are abundant with the similar properties to lithium, but there is a lack of suitable pseudocapacitive materials for sodium-based systems. The exploitation of pseudocapacitive materials for Na+ storage is urgent. This study, for the first time, reports a sonochemical approach, involving intercalation and ultrasonic exfoliation processes, to prepare composite K2Mo4O13/α-MoO3 (i.e., KMO) nanorods, and the possible applications of the KMO nanorods in Li+/Na+ pseudocapacitance were evaluated. The as-synthesized KMO possessed a uniform rod-like morphology formed by assembling nanoneedles (or nanobelts) with a large apparent aspect ratio of more than 10. Both in lithium ion batteries (LIBs) and sodium ion batteries (SIBs), the KMO nanorods exhibited efficient pseudocapacitance, which was not observed in the pristine MoO3–Na system. In SIBs, the as-synthesized KMO delivered a capacity of 895 mA h g−1 at 0.02 A g−1, which was much higher than that of the pristine MoO3–Na system. Moreover, it was found that the b values of KMO (i = avb, current i and scan rate v in the CV curves) were over 0.9 at potentials ranging from 1 V to 2 V in SIBs, indicating an obvious pseudocapacitive process. Benefiting from the layered K2Mo4O13 nanorods built with edge-shared distorted MoO6 octahedra, the Na+ ions with a larger size could be intercalated into the spaces between the double MoO6 plates. This sonochemical approach based on the intercalation and exfoliation chemistry opened a new path to prepare molybdenum-based nanostructures for superior Li+/Na+ pseudocapacitance applications on a large scale in a low-carbon and environment-friendly manner.


 Please wait while we load your content...
Please wait while we load your content...