Facile synthesis of porous hybrid materials based on Calix-3 dye and TiO2 for high photocatalytic water splitting performance with excellent stability†
Abstract
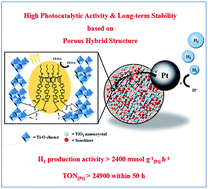
A highly robust hybrid material consisting of calixarene dye Calix-3 linked covalently to titanium dioxide, denoted as Calix-3-TiO2, is synthesized by a facile sol–gel method for the first time. The material not only has a micro/mesoporous structure and a large surface area, but also owns two clear band gaps: a narrow 1.7 eV and a wide 3.1 eV band gap, which can be ascribed to Calix-3 and Gel-TiO2, respectively. When Pt nanoparticles as co-catalysts are introduced onto the hybrid, the optimized Pt/Calix-3-TiO2 catalyst exhibits high hydrogen production activity (2444 mmol g−1 h−1 based on Pt mass) and performs with excellent stability even after recycling 10 times (a turnover number of 24 920 after 50 h), which is superior to that of the analogous sensitized Pt/Gel-TiO2/Calix-3 system and represents one of the highest among those reported heterogeneous catalytic systems under visible-light. The enhancement in efficiency and stability could be attributed to the more stable Ti–O–Calix-3 linkage and more efficient electron transfer between Calix-3 and TiO2. The micro/mesoporous TiO2 in the hybrid protects the dye, facilitates reactant/product transfer, and works as an electron transporting mediator and a reservoir simultaneously. The current study offers an inspiration for the design and application of new multi-functional organic–inorganic hybrid systems in efficient photocatalysis.


 Please wait while we load your content...
Please wait while we load your content...