Insights into Li/Ni ordering and surface reconstruction during synthesis of Ni-rich layered oxides†
Abstract
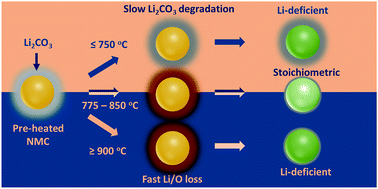
Nickel-rich layered transition metal oxides (NMCs) have been intensively studied as promising cathode candidates for next-generation Li-ion batteries, known for low cost and high theoretical capacity. However, the practical capacity of NMCs is largely determined by cationic ordering and has yet to be well controlled during synthesis, largely due to the complexity and non-equilibrium nature of the reactions occurring in the sintering process. In this work, high-energy synchrotron X-ray diffraction is employed to investigate the kinetic and thermodynamic aspects of cationic ordering during synthesis of LiNi0.7Mn0.15Co0.15O2 (NMC71515). It is found that cationic ordering in the bulk is coupled to surface reconstruction during synthesis, occurring concomitantly and both being greatly affected by Li2CO3 decomposition and Li loss at the particle surface. Through tuning the sintering temperature and time, highly ordered NMC71515 with high capacity and excellent rate capability is synthesized. The developed approach may be applied broadly to the synthesis of high-performance Ni-rich NMC and other cathode materials.


 Please wait while we load your content...
Please wait while we load your content...