Interaction of phospholipid vesicles with gemini surfactants of different lysine spacer lengths†
Abstract
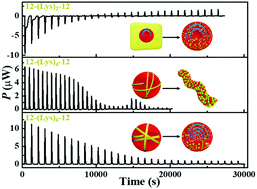
Peptide surfactants have shown many potential applications in biology and medicine; however, the mechanism of their interactions with biomembranes is still unclear. This work has studied the interactions of cationic peptide gemini surfactants based on lysine spacers (12-(Lys)n-12, n = 2, 4, and 6) with model biological membranes, which are represented by the vesicles separately formed by zwitterionic unsaturated phospholipid 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC), anionic unsaturated phospholipid 1,2-dioleoyl-sn-glycero-3-phospho-(1′-rac-glycerol) (DOPG, sodium salt) and the DOPC/DOPG (1 : 1) mixture. The experiment results show that the presence of negatively charged DOPG slightly affects the interaction manners of 12-(Lys)n-12 with the vesicles, while the interaction of 12-(Lys)2-12 with the phospholipid vesicles is significantly different from that of 12-(Lys)4-12 and 12-(Lys)6-12 with the vesicles. The binding strength decreases in the order of 12-(Lys)4-12 > 12-(Lys)6-12 > 12-(Lys)2-12. The 12-(Lys)4-12 surfactant solubilizes the DOPC vesicles, and makes the DOPC molecules join the surfactant stiff fibers and changes them into long and flexible wormlike micelles, while the 12-(Lys)6-12 and 12-(Lys)2-12 aggregates are disassembled by the DOPC vesicles, and the surfactant molecules join the DOPC vesicles and convert the unilamellar vesicles into multilamellar vesicles. This work should be helpful in understanding the interaction of peptide surfactants with phospholipid membranes.


 Please wait while we load your content...
Please wait while we load your content...