Oligo(ethylene glycol) side chain effect on the physical properties and molecular arrangement of oligothiophene–isoindigo based conjugated polymers†
Abstract
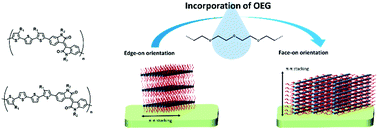
Oligo(ethylene glycol) (OEG) side chains are widely used in donor–acceptor conjugated polymers (D–A CPs) and enable the polymers to dissolve and be processed in environmentally friendly and cost-effective nonchlorinated solvents, such as water. However, the OEG effect on the physical properties of D–A CPs has not been thoroughly studied and sometimes the results are controversial. In this study, two oligothiophene–isoindigo based conjugated polymers, P3TI and P4TI, are selected as model polymers to investigate the OEG effect. PnTI has octyl side chains on the oligothiophene unit and 2-hexyldecyl side chains on the isoindigo unit. The replacement of an alkyl side chain with OEG not only changes the optical and thermal properties but also the molecular arrangements of the polymers such as π–π d-spacing, crystallinity, and packing orientation. The domination of the crystallization behavior changes from the oligothiophene unit to the isoindigo unit when the bulky alkyl group is replaced by the flexible and linear OEG. The packing changes from edge-on to face-on orientation. The results are intriguing and provide new insights into this class of polymers.


 Please wait while we load your content...
Please wait while we load your content...