Synthesis and characterisation of redox hydrogels based on stable nitroxide radicals†
Abstract
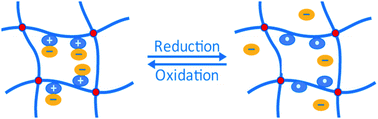
The principle of encapsulation/release of a guest molecule from stimuli responsive hydrogels (SRHs) is mainly realised with pH, temperature or light stimuli. However, only a limited number of redox responsive hydrogels have been investigated so far. We report here the development of a SRH that can release its guest molecule upon a redox stimulus. To obtain this redox hydrogel, we have introduced into the hydrogel the 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO) stable nitroxide radical, which can be reversibly oxidized into an oxoammonium cation (TEMPO+). Water solubility is provided by the presence of the (oligoethyleneglycol)methacrylate (OEGMA) comonomer. Electrochemical and mechanical characterization showed that those gels exhibit interesting physicochemical properties, making them very promising candidates for practical use in a wide range of applications.


 Please wait while we load your content...
Please wait while we load your content...