On the origin and evolution of the depletion zone in coffee stains†
Abstract
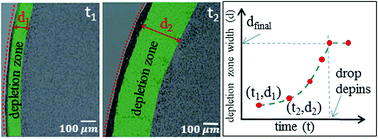
The variation in the concentration of surfactant molecules along the air–water interface of a drying sessile drop containing colloidal particle–surfactant mixtures is known to inhibit the formation of coffee stains. This also leads to the formation of particulate deposits with a region almost deprived of the particles, often called the depletion zone. The molecular size of the surface-active species used in such experiments poses limitations on the direct visualization of the build-up of surfactant molecules at the interface and how it correlates with the nucleation and growth kinetics of the depletion zone. We report a quantitative analysis of the origin and evolution of the depletion zone that forms in a drying sessile drop. By evaporating an aqueous sessile droplet containing a mixture of surface-active poly(N-isopropylacrylamide) (pNIPAM) microgels and polystyrene (PS) colloids, we establish that the concentration fluctuations of the PS particles along the air–water interface trigger a surface tension driven Marangoni flow along the interface. As a result of this, a depletion zone forms within the particulate deposit, which is analogous to the depletion zones observed for dried drops of particle– surfactant mixtures. The critical time at which the depletion zone forms correlates to the time at which surface tension-driven stress along the interface is maximum. Moreover, we show that the increase in the initial concentration of PS particles results in a decrease in the critical time. By using in situ video microscopy, we established that the rate at which the depletion zone grows is non-linear in time. The growth rate of the depletion zones for drops containing different concentrations of PS particles collapses into a master curve upon scaling with concentration and radius of the drop.


 Please wait while we load your content...
Please wait while we load your content...