Accelerative oxygen evolution by Cu-doping into Fe-Co oxides†
Abstract
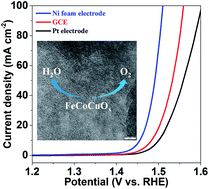
Development of cost-efficient, high-performance and robust earth-abundant electrocatalysts for the oxygen evolution reaction (OER) is a continuous challenge. Herein, we develop a facile sol–gel strategy to fabricate a ternary Fe-Co-Cu oxide for efficient electrocatalysis of water oxidation. Low overpotentials of 256, 271, and 226 mV at 10 mA cm−2 can be achieved over FeCoCuOx supported on a GCE, a Pt electrode, and Ni foam, respectively, in an alkaline electrolyte. Due to the enhanced charge transfer ability and electrochemically active surface area, trimetallic FeCoCuOx is more active than binary FeCoOx and among the most active OER catalysts to date.


 Please wait while we load your content...
Please wait while we load your content...