Novel CuO–Cu2O redox-induced self-assembly of hierarchical NiOOH@CuO–Cu2O/Co(OH)2 nanocomposite for efficient oxygen evolution reaction†
Abstract
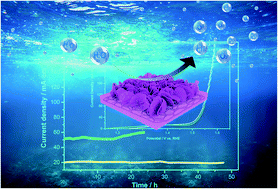
Low-cost and high-performance electrocatalysts are necessary for the oxygen evolution reaction (OER). Earth-abundant transition-metal-based catalysts are gradually replacing precious metal catalysts due to the scarcity of noble metals and their high cost. Herein, we report a facile method to synthesize an efficient electrode containing NiOOH and CuO–Cu2O components for the OER. Nickel foam (NF) was immersed in a mixed alcohol solution containing transition metal nitrate and urea via the solvothermal method at 100 °C. The CuO–Cu2O redox induced a valence change in nickel, and therefore catalyzed the self-assembly of NiOOH@CuO–Cu2O, which formed a tightly packed and strongly interacting hierarchical composite, contributing to the charge transfer. Besides, NiOOH@CuO–Cu2O was dispersed on the amorphous Co(OH)2 framework, which was grown in the network sheet of NF. The resulting hierarchical nanocomposite structure greatly increased the number of electron transfer channels. The improved conductivity of the electrode and active sites were conductive to the adsorption and diffusion of OH− from the electrolyte. As a working electrode, the obtained NCC-30 sample had a high catalytic performance for the OER with a low overpotential of 262 mV at a current density of 20 mA cm−2. It also showed long-term catalytic stability, where after 48 h i–t testing at 20 mA cm−2, its activity still retained 96% of the initial value and sustained the reaction for 15 h at 50 mA cm−2 in 1.0 M KOH. This work suggests that the CuO–Cu2O redox-induced in situ self-assembly of hierarchical NiOOH@CuO–Cu2O/Co(OH)2 nanocomposites grown in network sheets on NF is a new strategy for preparing efficient OER catalysts.


 Please wait while we load your content...
Please wait while we load your content...