The synergetic effect of acid and nickel sites on bifunctional MWW zeolite catalysts for ethylene oligomerization and aromatization†
Abstract
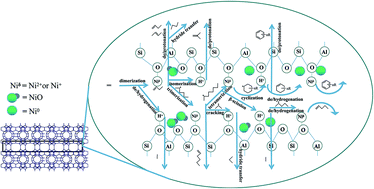
Corresponding H- and Ni-form MWW zeolites were synthesized and compared to investigate the influences of the acidity strength and the synergetic effect of bifunctional sites on the catalytic performance for ethylene oligomerization and aromatization. The acidities of ERB-1 and MCM-22 zeolites with an MWW structure were adjusted via deboronation and desilication to synthesize MCM-56 and ITQ-1. The physicochemical properties of the catalysts were measured via XRD, EDS, EPMA, N2 sorption, SEM, FT-IR, pyridine adsorption FT-IR, NH3-TPD, XPS and H2-TPR studies. The effects of the acid strength and Ni/acid site ratio on the product distribution were studied at 523 and 723 K, which are two typical optimal reaction temperatures for oligomerization and aromatization, respectively. According to the characterization and catalytic performance results from this series of catalyst zeolites with similar structures and different acid sites, a fundamental mechanism involving different elementary steps for ethylene oligomerization and aromatization was proposed to explain the synergetic effect of acid and Ni sites on bifunctional MWW zeolites. The synergetic effect impact relating to different acid strength MWW zeolite structures shows that Ni ions on weak acidity zeolites are superior for ethylene conversion and determine the Anderson–Schultz–Flory product distribution at 523 K, and strong acid sites on MCM-22 are preponderant for ethylene conversion and dominate the product distribution at 723 K. Ni species are not only favorable for the dimerization of α-olefins at 523 K, but also for dehydrogenation, leading to the generation of aromatics at 723 K. Increasing zeolite acidity strength produces an obviously positive effect on the production of aromatics.


 Please wait while we load your content...
Please wait while we load your content...