Carbon-based catalysts for electrochemical CO2 reduction
Abstract
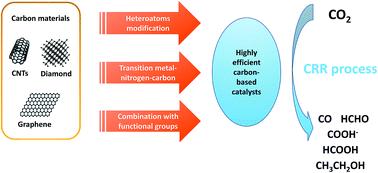
Fossil fuel energy gives rise to various global issues including aggravated greenhouse gas carbon dioxide (CO2) emission, which deteriorates the surrounding environment resulting in global warming and melting glaciers. Electrochemical CO2 reduction is regarded as a promising method to convert CO2 back into valuable chemicals and fuels, in particular when electricity is generated from renewable sources such as sunlight and wind. Carbonaceous materials are considered to be promising catalysts for this purpose due to their excellent electroconductivity, high surface area and low cost. Normally, pure carbon materials have no activity because of the electroneutrality of carbon atoms. To design carbonaceous catalysts with superior activity, various efficient and convenient strategies have been reported in recent years. In this review, we first provide a sketch about doping heteroatoms into a carbon structure which can break the electroneutrality of carbon frameworks and enhance the adsorption capacity of the CO2 intermediate. Furthermore, a transition metal–nitrogen–carbon system is summarized as a highly efficient structure for CO2 reduction. In addition, a series of reports representative of carbon substrates combined with different types of functional groups such as metal particles, metal oxides, metal complexes and even metal-free functional groups are reviewed. Finally, we highlight some potential limitations for the reported catalytic systems which hopefully can direct researchers to investigate new carbon-based catalysts and synthesis strategies for the development of electrochemical CO2 reduction in the future.


 Please wait while we load your content...
Please wait while we load your content...