Diperovskite (NH4)3FeF6/graphene nanocomposites for superior Na-ion storage†
Abstract
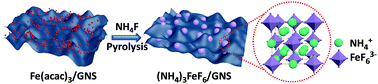
Transition metal fluorides (TMFs) have attracted much attention as electrode materials for sodium-ion batteries (SIBs) due to their abundance, low cost, and high specific capacity. However, intrinsic low electronic conductivity and high resistance to Na ion diffusion impede the applications of TMFs in SIBs. Herein, diperovskite-type (NH4)3FeF6/graphene nanosheet ((NH4)3FeF6/GNS) composites were successfully synthesized using a facile co-pyrolysis approach from ferric acetylacetonate (Fe(acac)3) and ammonium fluoride (NH4F) with the addition of GNS. The pure diperovskite phase of (NH4)3FeF6 was generated at reaction temperatures ranging from 150–300 °C using a high NH4F/Fe(acac)3 molar ratio. The stable diperovskite structure, bearing an open three-dimensional framework, was favourable for the fast storage of large-sized Na ions. Therefore, the (NH4)3FeF6/GNS composites exhibited a high capacity of 487.8 mA h g−1 at 0.1 A g−1 and excellent rate performances as anode materials for SIBs. The specific capacities of the (NH4)3FeF6/GNS composites were up to 257.8, 219.4, and 180.6 mA h g−1 at 2.0, 5.0, and 10.0 A g−1, respectively. Even at 20.0 A g−1 (approx. 58C), the (NH4)3FeF6/GNS composites still delivered a specific capacity of 149.5 mA h g−1 and exhibited a long lifespan up to 3000 cycles. Therefore, the (NH4)3FeF6/GNS composites are potential anode materials for SIBs.


 Please wait while we load your content...
Please wait while we load your content...