An organic ligand promoting the electrocatalytic activity of cobalt oxide for the hydrogen evolution reaction†
Abstract
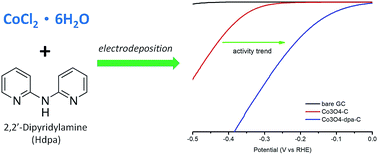
In electrocatalytic water splitting, active, stable and low-cost hydrogen evolution electrocatalysts play key roles in the conversion and storage of renewable energy sources. Here we report that the incorporation of an organic ligand (2,2′-dipyridylamine (Hdpa)) into Co3O4 dramatically enhanced its electrocatalytic activity for the hydrogen evolution reaction. The co-electrodeposited Co3O4-dpa-C nanosheets on a glassy carbon electrode exhibited a low overpotential of 54 mV to achieve a current density of 10 mA cm−2 for the HER, much lower than that of pure Co3O4 (324 mV at 10 mA cm−2). These results indicate that the molecular Co complex anchored on the surface of Co3O4 acts as the catalytic center and cooperatively promotes the hydrogen evolution reaction at the interface of the Co3O4 support under alkaline conditions.
- This article is part of the themed collection: 2019 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...