Solvent-controlled solid-electrolyte interphase layer composition of a high performance Li4Ti5O12 anode for Na-ion battery applications†
Abstract
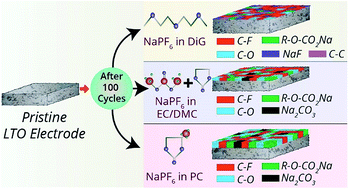
Intrigued by the unlimited availability, low cost and high redox potential of sodium, the rechargeable sodium ion battery (SIB) has attracted significant research interest as a supplement to lithium ion batteries. However, an optimized electrolyte for SIB has still not been developed, which in turn affects the performance of the electrode materials. Herein, we have elucidated the influence of electrolyte formulations in the design of a high performance lithium titanate (LTO) anode for sodium ion battery applications. NaPF6 salt in three solvent formulations (diglyme (DiG), propylene carbonate (PC), and ethylene carbonate-dimethyl carbonate (EC/DMC)) were explored to investigate the electrochemical performance of solvothermally synthesized surface engineered LTO samples. An unprecedented rate capability (up to 300C), cycling stability (1000 cycles) and wide temperature range of operation (−10 °C to 55 °C) were demonstrated by the LTO–diglyme based electrode, the highest rate performance reported so far for LTO-based sodium ion battery anodes. Detailed surface chemical analysis via ex situ XPS measurements was performed on the pristine and cycled electrodes in each electrolyte to rationalize the electrode's performance and realize the solid-electrolyte interphase (SEI) evolution and its composition. XPS confirmed the formation of a thin, protective SEI layer in DiG based electrolyte composed of mixed organic/inorganic components resulting in the excellent rate and cycling capabilities. In contrast, PC- and EC/DMC-based electrolytes formed a relatively thicker, unstable, organic-rich SEI layer, leading to inferior electrochemical performance.
- This article is part of the themed collection: 2019 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...