Effects of the fabrication strategy on the catalytic performances of Co–Ni bimetal ordered mesoporous catalysts toward CO2 methanation†
Abstract
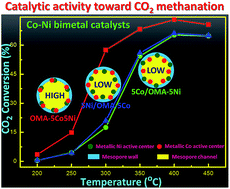
The interfacial effect between different metallic active centers can greatly influence their synergistic effect, further affecting their catalytic performance. Therefore, Co–Ni bimetal based ordered mesoporous metal oxide catalysts with different interfacial properties have been prepared by a one-pot evaporation induced self-assembly (EISA) strategy as well as an incipient impregnation method. The obtained catalysts with large specific surface areas, large pore volumes, and narrow pore size distributions were investigated as catalysts toward CO2 methanation. It was found that the Co–Ni bimetal catalyst synthesized by the one-pot EISA strategy displayed a much higher catalytic activity in CO2 conversion than the counterparts prepared by the incipient impregnation method, especially in a low reaction temperature region. The reason for this could be attributed to the difference in their interfacial properties based on XPS and H2-TPR characterization studies, which greatly affected the catalytic activities. Besides, all the investigated catalysts showed excellent catalytic stabilities without deactivation after 50 h time on stream owing to the confinement effect of the mesoporous framework and mesoporous channel, which could effectively suppress the thermal sintering of the metallic active centers. Therefore, the Co–Ni based bimetal ordered mesoporous catalysts constructed by the one-pot EISA strategy could be considered as a potential efficient catalyst candidate for CO2 methanation.


 Please wait while we load your content...
Please wait while we load your content...