Graphene edge-enhanced anchoring of the well-exposed cobalt clusters via strong chemical bonding for accelerating the oxygen reduction reaction†
Abstract
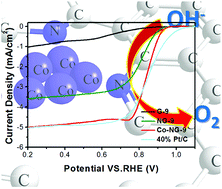
Efficient anchoring of transitional metals on supporting materials with well-exposed metal atoms and tuned chemical bonds between them is crucially important for the design of high-performance electrocatalysts. In this study, we adopted a strategy of using defective carbon nanotubes to synthesize holey graphene oxide with tunable basal plane hole size. Via the in situ formation of cobalt clusters during the process of doping of nitrogen into holey graphene oxide, the obtained composite showed strong chemical bonding of highly exposed cobalt with nitrogen and carbon on the nitrogen-doped holey graphene. It was found that the basal plane hole of graphene was beneficial for the deposition of cobalt; detailed analysis showed that rich defects on the basal plane favored the doping of pyridinic nitrogen and graphitic nitrogen and enhanced the chemical bonding of cobalt with nitrogen; this improved the four-electron transfer selectivity of the ORR; thus, the effectively enhanced catalytic activity of composites was comparable to those of the commercial Pt/C catalyst; moreover, these composites exhibited superior stability than the commercial Pt/C catalyst.


 Please wait while we load your content...
Please wait while we load your content...