Enhanced water splitting through two-step photoexcitation by sunlight using tantalum/nitrogen-codoped rutile titania as a water oxidation photocatalyst†
Abstract
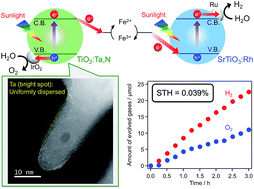
Rutile TiO2 codoped with tantalum and nitrogen (TiO2:Ta,N) was assessed as a water oxidation photocatalyst for Z-scheme water splitting driven by visible light. This material was prepared by thermal ammonolysis of TiO2:Ta with dry NH3 at 773 K, while samples of the oxide precursor were synthesized using a microwave-assisted solvothermal technique, applying various conditions. The photocatalytic activity of the TiO2:Ta,N during water oxidation to O2 from an aqueous FeCl3 solution was found to be greatly affected by the synthesis parameters. The rate of O2 evolution was increased upon increasing the level of Ta doping in conjunction with a highly-crystalized TiO2:Ta precursor resulting from applying a higher temperature during synthesis of the oxide. IrO2 loading of the TiO2:Ta,N photocatalyst also improved the O2 evolution activity. The optimized IrO2/TiO2:Ta,N photocatalyst was applied to a Z-scheme water splitting system in combination with Ru/SrTiO3:Rh and in the presence of redox mediators (Fe3+/2+ or [Co(bpy)3]3+/2+). Under AM1.5G simulated sunlight, this system exhibited a maximum solar-to-hydrogen energy conversion efficiency of 0.039%, which was nearly twice as high as the previously reported system with the use of RuO2/TiO2:Ta,N (0.021%).


 Please wait while we load your content...
Please wait while we load your content...