Environment-friendly, co-catalyst- and solvent-free fixation of CO2 using an ionic zinc(ii)–porphyrin complex immobilized in porous metal–organic frameworks†
Abstract
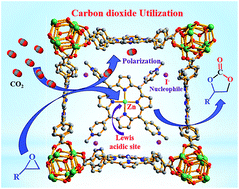
Herein an ionic Zn(II)–porphyrin complex, [Zn(II)NMeTPyP]4+[I−]4 (Zn(II)NMeTPyP = 5,10,15,20-tetrakis(1-methylpyridinium-4′-yl) zinc(II) porphyrin), was immobilized in a porous metal–organic framework, PCN-224. The resulting [Zn(II)NMeTPyP]4+[I−]4@PCN-224 hybrid was thoroughly characterized by PXRD, UV-vis, FTIR, MP-AES, SEM-EDS and BET analysis. Further, DFT calculations confirmed the incorporation of the [Zn(II)NMeTPyP]4+[I−]4 complex in the 1D channels of PCN-224 owing to appropriate matching of the pore size with the size of the complex and stabilization via π–π interactions. Interestingly, the [Zn(II)NMeTPyP]4+[I−]4@PCN-224 hybrid showed high affinity for CO2 with a high heat of adsorption (Qst) value of 40.5 kJ mol−1 compared to the pristine MOF, PCN-224, with a Qst value of 36.8 kJ mol−1. Remarkably, the hybrid material acts as an efficient environment-friendly, co-catalyst- and solvent-free heterogeneous catalyst for chemical fixation of CO2 to generate cyclic carbonates with high yield and selectivity due to synergistic cooperation between the complex and the framework.
- This article is part of the themed collection: 2019 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...