Ni2P2O7 microsheets as efficient Bi-functional electrocatalysts for water splitting application†
Abstract
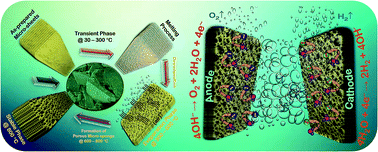
The prospect of promoting new electrode materials to substitute noble metal electrocatalysts appears to be a boundless challenge. Hence, identifying appropriate materials with enhanced structural and fascinating morphological properties that can compete with the electrocatalytic properties of noble electrocatalysts is necessary. Herein, we report the electrocatalytic properties of nickel pyrophosphate (Ni2P2O7) microsheets with a novel architecture, which were synthesized via a facile hydrothermal route followed by thermal treatment. The prepared single-phase crystalline Ni2P2O7 with a unique morphology was found to be an active bi-functional electrocatalyst for efficient water splitting systems. The Ni2P2O7 microsheets with a highly porous surface promoted an enhancement in adsorption sites for the oxygen evolution reaction with a small overpotential of 210 mV at 10 mA cm−2. Also, the dominant characteristics of the metal pyrophosphates boosted the catalytic activity for the hydrogen evolution reaction with a nominal overpotential requirement of 185 mV at the same current density. The uniformly distributed constitutional elements with an improved porous structure and enhanced surface roughness factor resulted in superior electrocatalytic activity for the prepared Ni2P2O7 electrode. Convincingly, a lab-scale water electrolyzer was fabricated and the tested electrode gave a current density of 10 mA cm−2 with a low cell voltage of 1.61 V. Hence, the Ni2P2O7 material with excellent electrocatalytic properties and durability can be promoted as a potential electrode material.


 Please wait while we load your content...
Please wait while we load your content...