Diffusion controlled porous WO3 thin film photoanodes for efficient solar-driven photoelectrochemical permanganic acid production†
Abstract
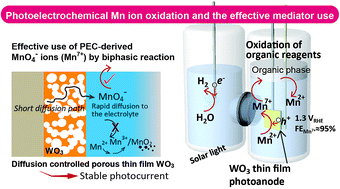
Photoelectrochemical (PEC) oxidation of the divalent manganese ion to the permanganate ion was achieved by using porous WO3 thin film photoanodes in sulfuric acid electrolytes under simulated sunlight. In this reaction system, the subsequent reaction of the evolved MnO4− (Mn7+) with residual Mn2+ caused deterioration of photoanodes by Mn solid deposit formation. However, we realized stable PEC reaction by diffusion control of the evolved Mn7+ in the pores of the photoanodes. We also confirmed that the PEC reaction for the evolution of Mn7+ proceeded intrinsically very efficiently, and the faradaic efficiency reached 94.8% in an electrolyte containing 1.0 mM Mn2+ and 1.0 M H2SO4 at a low bias potential voltage of 1.3 VRHE. To use the photoelectrochemically evolved Mn7+ in sulfuric acid solutions effectively, a biphasic reaction involving the oxidation of ethylbenzene was carried out. The ethylbenzene added to the anode cell was converted to acetophenone without any byproducts, and this conversion occurred concurrently with the PEC reaction that produced Mn7+. At the cathode, evolution of hydrogen occurred with 95–99% faradaic efficiency. Thus, we have successfully demonstrated dual benefits of this reaction system: the production of high-value-added organic reagents mediated by the PEC-derived Mn7+ and the production of solar hydrogen.


 Please wait while we load your content...
Please wait while we load your content...