A silicon-doped iridium electrode prepared by magnetron-sputtering as an advanced electrocatalyst for overall water splitting in acidic media†
Abstract
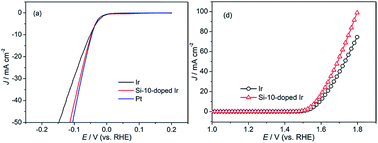
With increasing demands for clean and renewable energy, electrocatalytic water splitting is considered as the most promising procedure of hydrogen production. Pt is the best catalyst for the hydrogen evolution reaction (HER). However, the low oxygen evolution reaction (OER) activity of Pt prevents it from becoming a bifunctional catalyst in practical application. Ir-based electrocatalysts with good OER activities are expected to become the most promising bifunctional catalysts once their HER activities are improved. Herein, we report a simple synthesis of a Si-doped Ir electrode using magnetron sputtering. The physical and electrochemical characterization of the materials is achieved by scanning electron microscopy (SEM), atomic force microscopy (AFM), X-ray fluorescence (XRF), X-ray diffraction (XRD) and X-ray photoelectron spectrometry (XPS). The electrochemical catalytic activity of the Si-doped Ir electrode toward the HER is demonstrated for the first time. Cyclic voltammetry (CV) curves reveal that the addition of Si can improve the utilization rate of Ir and result in a large electrochemical surface area (ESA). The overpotential of the Si-doped Ir electrode for the HER (η = 114 mV, 50 mA cm−2) is obviously lower than that of an Ir electrode (η = 148 mV, 50 mA cm−2) and very close to that of a Pt electrode (η = 106 mV, 50 mA cm−2). The mass specific activity of the Si-doped Ir electrode exceeds that of an Ir electrode by a factor of ca. 2 at an overpotential of η = 200 mV. The superior HER activity of the Si-doped Ir electrode could be attributed to the electronic structure modification of Ir by the interaction effect with Si as studied through XPS analysis. Moreover, the Tafel slope of 36.5 mV dec−1 suggests that the mechanism for the Si-doped Ir electrode-catalyzed HER is Volmer–Tafel, in which the recombination of two adsorbed hydrogen atoms is the rate-determining step. In addition, the OER activity of the Si-doped Ir electrode outperforms that of an Ir electrode, which enables the Si-doped Ir electrode to be used as a bifunctional catalyst for overall water electrolysis.


 Please wait while we load your content...
Please wait while we load your content...