Fast microwave-induced synthesis of solid cobalt hydroxide nanorods and their thermal conversion into porous cobalt oxide nanorods for efficient oxygen evolution reaction†
Abstract
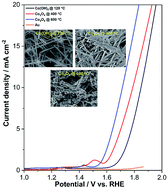
Oxygen evolution reaction (OER) in water splitting is one of the most critical and more demanding half-reactions in electrochemical devices; therefore, the design of highly efficient and nonprecious metal-based electrocatalysts is required. Herein, we presented the fast microwave synthesis of the solid nanorods of Co(OH)2 and their conversion into interconnected porous nanorods of Co3O4 by high-temperature calcination for exposing active sites, which offered a high surface area; this resulted in faster exchange of intermediates and more efficient electron transfer for enhanced OER activities in alkaline solutions. The as-synthesized solid Co(OH)2 and calcined porous Co3O4 nanorod catalysts were characterized by scanning and transmission electron microscopies (SEM and TEM), powder X-ray diffraction (PXRD), and X-ray photoelectron spectroscopy (XPS); finally, the OER performance was investigated for water oxidation. FE-SEM and HR-TEM images revealed the formation of solid nanorods of as-synthesized Co(OH)2, which were broken and converted into porous interconnected nanorods after calcination at a high temperature. In 0.1 M KOH aqueous electrolyte, solid Co(OH)2 @ 120 °C nanorods did not have favorable OER activity, while the Co3O4 @ 600 °C electrocatalyst exhibited a low onset potential (1.55 V vs. RHE) and Tafel slope (49 mV dec−1), maintaining robust stability for more than 18 h with negligible fading. This is mainly because the interconnected porous nanorod Co3O4 @ 600 °C catalyst possessed rich active boundary sites and short paths for charge/mass transport (higher reactivity of active sites) than Co3O4 @ 400 °C and Co(OH)2 @ 120 °C. Such high catalytic activity was attributed to the sufficient exposure of the accessible active sites of porous Co3O4 electrocatalysts.
- This article is part of the themed collection: 2019 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...